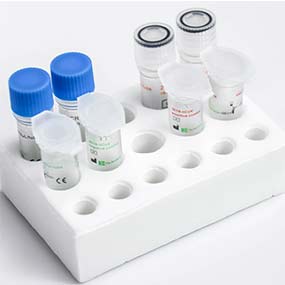
COVID-19 is a highly transmissible and pathogenic disease caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and has now threatened human health and overwhelmed many healthcare systems.
WHO announced the COVID-19 outbreak as a pandemic on March 11, 2020, due to the rapid increase in the number of cases around the world.