There are approximately 58 million people infected with the hepatitis C virus, the new infection is increased by around 1.5 million per year. In 2019, WHO estimated that about 290 000 people died from hepatitis C aggravation. In this article, we will discuss the importance of HCV RNA detection in the treatment of hepatitis C infection.
Hepatitis C is caused by the virus hepatitis C virus (HCV) which is an enveloped RNA virus with a positive-sense single-stranded structure. HCV attacks liver cells and leads to hepatic injury, it also escapes innate and adaptive immunity and causes chronic infections. Currently, all the known HCV genotypes are hepatotropic and pathogenic, despite the differences in nucleotide sequences between HCV genotypes. HCV has many genotypes, they are various in nucleotide sequence. Difference genotypes are various in infectivity and pathogenicity, which has an impact on the rate of progression of cirrhosis and the risk of HCC. The heterogeneity of HCV would also lead to different responses to anti-viral therapy.
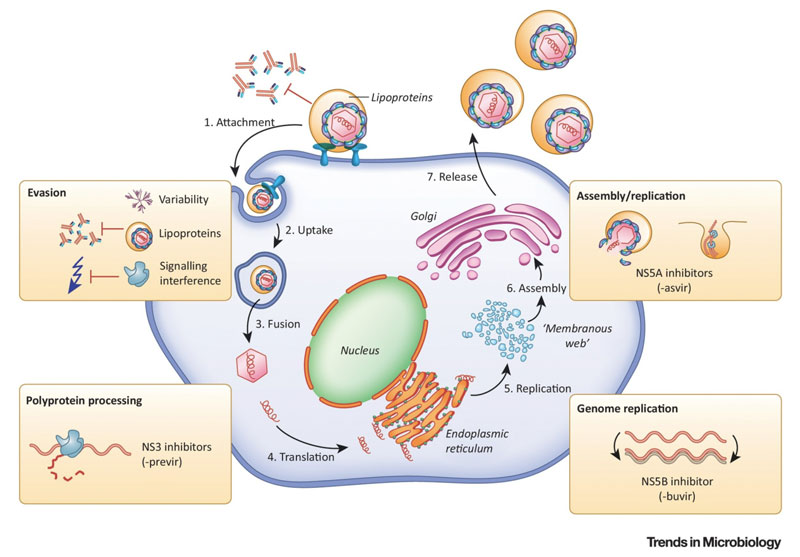
Source: https://www.cell.com/trends/microbiology/fulltext/S0966-842X(19)30013-7
Cirrhosis and hepatocellular carcinoma are the lethal factors for HCV-infected patients. Acute hepatitis C infections usually occur asymptomatically, and around 30% of the infected patient can eradicate the virus spontaneously within 6 months without treatment. However, about 70% of infected cases failed to clear the virus within 6 months and the patient enter the chronic phase. The virus attacks the liver cells leading to cirrhosis, if the cirrhosis gets aggravation it will develop into Hepatocellular carcinoma.
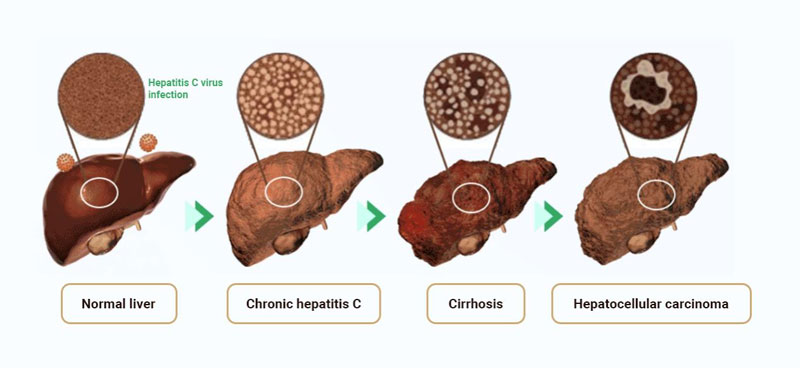
HCV RNA is usually detected by reverse transcription-polymerase chain reaction(RT-PCR) to evaluate the quantification of HCV RNA levels in clinical practice. The HCV PCR assay is very sensitive with a broad dynamic range of quantification and may prevent residual contamination. Serum is an ideal specimen for an HCV RNA quantification test, HCV RNA levels tested by NATs are usually used as the confirmatory tool to diagnose CHC infection. HCV RNA detection is also an effective tool for determining the viral load both before and during antiviral treatments.
Accurate diagnosis is important in hepatitis treatment, Daan Gene dedicates to providing reliable and efficient solutions for viral hepatitis diagnosis.
Feature of DaAn Gene HCV RNA PCR Qualitative Test Kit
Easy and fast: Simple experimental operation, results are available within two hours.
Reliable results: No cross-reactivity with other pathogens.
High sensitivity: LoD 20 IU/mL
Internal control:Monitor the whole process.
>> Learn More about DaAn Gene HCV PCR Test Kit
Reference:
1. Li HC, Lo SY. Hepatitis C virus: Virology, diagnosis and treatment. World J Hepatol. 2015 Jun 8;7(10):1377-89. doi: 10.4254/wjh.v7.i10.1377. PMID: 26052383; PMCID: PMC4450201.
2. https://www.cell.com/trends/microbiology/fulltext/S0966-842X(19)30013-7
3. http://www.hepctrust.org.uk/information/impact-hepatitis-c-liver/progression-hepatitis-c
4. https://www.who.int/news-room/fact-sheets/detail/hepatitis-c





